The Birmingham Mid Head Resection (BMHR)
N.B. The Birmingham Mid-Head Resection is currently no longer in use by Professor McMinn. However, due to its existing positive results in our patients, the device is being developed as part of future resurfacing device designs. For more information, please contact The McMinn Centre on (+44) 0121 455 0411 or via email at enquiries@mcminncentre.co.uk.
Click on the thumbnails for enlarged view
 The BMHR (Birmingham Mid Head Resection) is an alternative, bone-conserving hip device for young and active patients who are unsuitable for a BHR. The BMHR (Birmingham Mid Head Resection) is an alternative, bone-conserving hip device for young and active patients who are unsuitable for a BHR.
The BHR procedure is typically unsuitable for patients with avascular necrosis (AVN), large femoral cysts, or patients with anatomical difficulties such as hip dysplasia or Perthes disease. With these diagnoses, patients would often suffer failure if treated with a BHR because the quality of their femoral bone is compromised, or they simply don’t have enough bone available to securely implant a BHR.
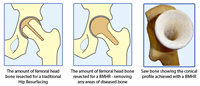 For patients with these types of hip conditions, Professor McMinn has developed another bone-conserving hip device, the BMHR (sometimes called the “hip resurfacing plus”). The BMHR is specifically designed as an alternative surgery for young and active patients who would otherwise be forced to have a total hip replacement (THR) in the absence of a hip resurfacing. Like the BHR, the BMHR procedure restores the patient’s natural anatomy and allows them to be active following surgery. For patients with these types of hip conditions, Professor McMinn has developed another bone-conserving hip device, the BMHR (sometimes called the “hip resurfacing plus”). The BMHR is specifically designed as an alternative surgery for young and active patients who would otherwise be forced to have a total hip replacement (THR) in the absence of a hip resurfacing. Like the BHR, the BMHR procedure restores the patient’s natural anatomy and allows them to be active following surgery.
During the BMHR operation, the femoral head (ball joint) is resected through the middle, thereby removing the diseased or necrotic bone. The remaining femoral bone is then prepared into a conical shape and a matching stem is tapped into place (without the need for cement or screws). The stem is then fitted with a large diameter modular head (matching the patient’s natural anatomy), which fits into the same acetabular cup as used for a BHR.
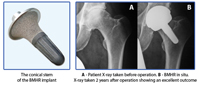 The BMHR procedure is specifically designed to remove any diseased or necrotic bone, and then “fill in” the femur with a chunky, conical stem. Testing shows that the conical stem replicates the physiological loading of the native femur, preventing any stress-shielding bone loss in the long term. The BMHR procedure is specifically designed to remove any diseased or necrotic bone, and then “fill in” the femur with a chunky, conical stem. Testing shows that the conical stem replicates the physiological loading of the native femur, preventing any stress-shielding bone loss in the long term.
The BMHR is made up of three components - the femoral stem, a modular head and an acetabular cup. The modular head and cup are manufactured from as-cast Cobalt Chrome which has a proven clinical history of over 40 years. The BMHR stem is manufactured from Titanium Alloy which also has a long history of clinical success with orthopaedic devices.
The BMHR is built on the clinical success of the BHR, offering all the advantages of a hip resurfacing - a good range of movement, excellent stability, reduced risk of dislocation and better revision options than a THR.
As the pioneer behind the device, Mr McMinn is one of the few experienced surgeons in the world who can offer the BMHR procedure to his patients.
The Ceramic BMHR
For the small number of patients with metal allergies, Professor McMinn has started using a BMHR stem fitted with a custom-made ceramic modular head. This device is a "Ceramic-on-Metal" (CoM) articulation. Early results with this type of CoM device show significantly lower metal wear than with any metal-on-metal hip replacement, making it a safer procedure for patients with metal allergies. You can find out more about this option by clicking here.

|